How UV Technology Protects Against Waterborne Pathogens in Pharmaceutical Applications
Posted on March 5, 2026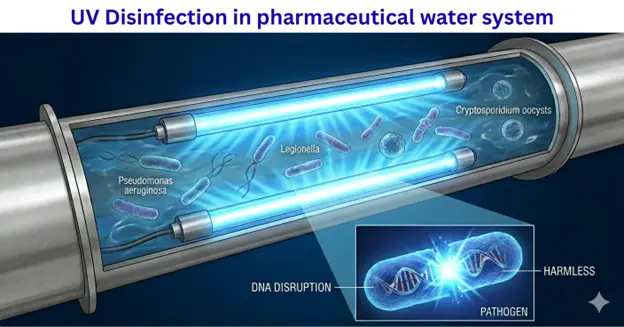
Introduction
Water purity is extremely important for the pharmaceutical industry. Each drop used in manufacturing must meet high standards, or the whole process is at risk.
Waterborne pathogens are a real threat. These tiny germs can contaminate products and make them unsafe for users. If this happens, it can lead to PR threat, recalls, cause health risks, and result in failure to meet regulations.
To manage this risk, more pharmaceutical companies are choosing UV Technology. This modern solution controls pathogens effectively and keeps water systems safe, without using harmful chemicals.
The Role of Water in Pharmaceutical Manufacturing
Water is used at every stage in pharma plants—for drug formulations, cleaning, and sterilization. Because water is so essential, its quality must meet tough standards.
Pharmaceutical manufacturing water must comply with standards like IP – Indian Pharmacopeia, BP (British Pharmacopoeia), and USP (United States Pharmacopeia) and guidelines from regulatory agencies such as CDSCO (Central Drugs Standard Control Organisation) and the FDA (Food and Drug Administration). These ensure that water used in making medicines is free from contamination and safe for all processes.
Basic water treatment isn’t enough to meet such strict requirements. manufacturers must install advanced systems to make sure their water always meets IP and other compliance standards.
The Threat of Waterborne Pathogens
Waterborne pathogens are microbes—like bacteria, viruses, and parasites—that can survive in water. In pharmaceutical manufacturing, even a tiny amount can quickly multiply and contaminate medicines.
Common troublemakers include Pseudomonas aeruginosa, Legionella, and Cryptosporidium. These are not always easy to detect and can survive in tanks and piping.
Traditional disinfection, like chlorine, doesn’t always work. Some pathogens resist it, and leftover chemicals are not allowed in pharmaceutical water as per regulatory norms. This is why new approaches are often needed in the market.
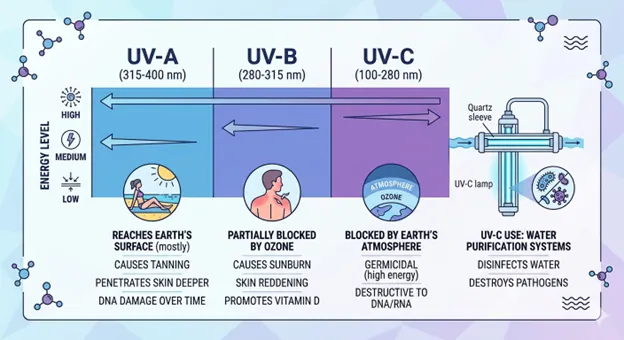
How UV-C Technology Safely Neutralizes Waterborne Pathogens
UV disinfection uses UV-C light to neutralize harmful microbes in water. The light disrupts the DNA of bacteria and viruses so they cannot reproduce.
This process does not use any chemicals, which is important for meeting IP and CDSCO requirements. UV is also proven to be especially effective against tough microbes like Cryptosporidium that commonly challenge pharmaceutical plants.
Benefits of UV Technology in Pharmaceutical Applications
Chemical-Free Disinfection
UV systems add nothing to the water. This means there are no chemical residues, helping companies stay within safety and purity regulations.
High Efficacy
UV works quickly to control bacteria, viruses, and parasites—even those resistant to other treatments.
Keeps Water Quality Intact
With UV, water’s natural properties aren’t changed. It keeps its taste, pH, and purity—all essential for medicine-making under law.
Cost Savings
Using UV technology can lower ongoing costs by reducing the need for chemicals and cutting down on system downtime from contamination.
Sustainability
UV supports water-saving and green initiatives. It also helps companies meet growing eco-friendly requirements by reducing chemical waste.
Real-World Applications of UV in Pharma
More pharmaceutical companies are successfully using UV technology to stay compliant with both local and international standards.
Choosing the Right UV System for Pharmaceutical Needs
Ensure the system fits your existing setup in terms of flow rates and piping sizes. Features such as UV intensity monitoring are useful to meet regulatory audits and checks.
Working with experienced providers like Alfaa UV ensures you get a system that fits your unique needs and regulatory demands.
Conclusion
UV technology helps pharmaceutical manufacturers protect their water from dangerous pathogens. It shields your products and users, and supports compliance with IP and other standards.
The benefits are clear: it is safe, effective, and chemical-free. It also supports sustainability and cost savings for companies.
For the best way to ensure the purest water, explore Alfaa UV’s advanced solutions made for the pharma sector.